NOTE: This is a case study using a new precision paradigm on how to assess risks of environmental chemicals by reliance on the pharmaceutical development process and its science. More about that new paradigm at: Precision evaluation of environmental chemical risk assessment
The development of a cancer’s resistance to chemotherapy has become a disheartening and all-too-common phenomenon. A wide variety of strategies and drugs have been developed along with new fundamental ways to look at this frequently deadly problem: Cancer therapy: an evolved approach1.
Into this mix come pharmaceuticals targeting the molecular interactions associated with chemotherapy resistance.
Ironically, the very intravenous bags and tubing used for chemotherapy along with other medical devices can leach Bisphenol A (BPA): a chemical that directly counteracts the same pharmaceutical chemical described below which is used to combat resistance2.
Previous studies have linked BPA to chemoresistance but have not been able to specify the precise molecular mechanism3.
Other studies have found that BPA interferes with chemotherapy, but those have been dismissed as irrelevant because they were in vitro investigations and not directly applicable4.
These criticisms have diminished the perceived risk of BPA.
However, a new method of assessing environmental chemical risk by using data from pharmaceutical trials can pinpoint precise molecular mechanisms and improve risk assessments.
Already-approved chemo resistance drug entering new research trials
One such pharmaceutical recently announced the start of stage II clinical trials targeting the Hsp27 heat shock protein.
That announcement4 stated that, “RedHill Biopharma Ltd … is collaborating on research with Leipzig-based Fraunhofer Institute for Cell Therapy and Immunology (IZI), for the evaluation of RedHill’s Phase 2-stage oncology drug candidate, RP101. IZI is a research unit of the Fraunhofer Society.”
According to RedHill Biopharma, RP101 has been granted Orphan Drug designation for the adjunct treatment of pancreatic cancer by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA)6.
RedHill’s statement said that, “RP101 binds to Hsp27, a chaperone protein which is found in abnormally high levels in cancer cells, and inhibits its activity. The over-expression of Hsp27, which results in the amplification of a multidrug-resistance (MDR) gene, has been linked to tumor resistance to cytotoxic drugs and the development of metastasis.”
In addition to the RedHill effort, Hsp27 has been the subject of a number of published, peer-reviewed studies describing the role played by Hsp27 in notable cancer-promotion roles.
This ad-free article is made possible by the financial support of the
Center for Research on Environmental Chemicals in Humans: a 501(c)(3) non-profit.
Please consider making a tax-deductible donation for continued biomedical research.
Bisphenol A and Cancer
As noted above, current equipment and supplies used to administer chemotherapy agents such as RP101 are made from plastics containing Bisphenol A. Now comes a major study7 whose results find that BPA in environmentally relevant concentrations over-expresses Hsp27.
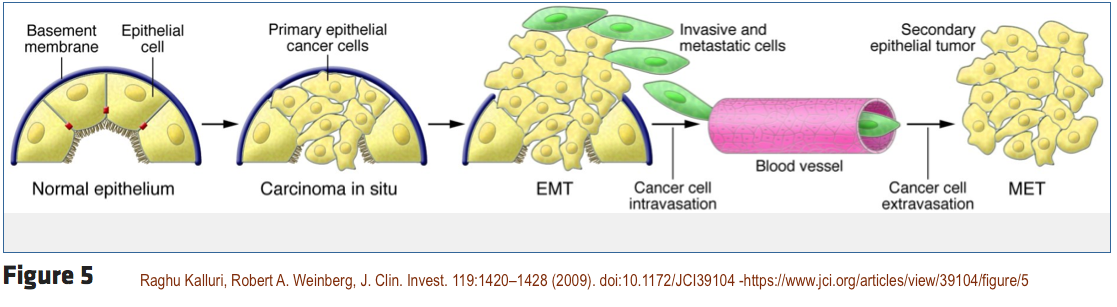
That study by Chen et al., found that “the xenoestrogen BPA at nanomolar and greater concentrations modulates the protein profiles and promotes the metastasis of colorectal cancer cells via induction of EMT.” EMT stands for “Epithelial-Mesenchymal Transition” and is described below in greater detail.
“Greater expression of Hsp27 protein has previously been reported in colon cancer cells and associated with poor prognosis (Thanner et al. 2005). The fact that exposure to the oral equivalent concentration of BPA resulted in significantly greater expression of Hsp27 protein might indicate that low dose of BPA can also promote tumorigenesis and development of colorectal cancer.”
The cross-referencing of the findings by Chen et al. with the clinical trials of RP101 establish a probable mechanism between BPA and cancer. Given the exceptional rigor of government-mandated pharmaceutical trials, this cross-link between lab studies and drug trials indicates the need to require more thorough testing of BPA using the most current investigation techniques.
EMT — Epithelial-Mesenchymal Transitions
EMT — Epithelial-Mesenchymal Transitions — are vital processes in embryo and organ growth that have been hijacked by cancers. EMT changes cells from stable tissue where they are attached to each other and gives provides them with a greater ability to invade neighboring cells.
EMT also reduces normal programmed cell death (apoptosis) and allows cells to detach and float freely in the bloodstream which promotes metastasis.
Significantly, the EMT process encompasses three of the original six “Hallmarks of Cancer” described in a landmark paper by that title which was published in the top-tier journal Cell. A quick, non-technical summary of that paper is available here: The Hallmarks of Cancer in a Nutshell.
It is notable that Chen et al. found that — in addition to its EMT effects on Hsp27 — BPA in nano- and micromolar concentrations is active in disrupting more than a dozen other molecular pathways that have significant impacts on multiple other cancer promoters.
Finally, the effects of Hsp27 and other heat shock proteins on numerous types of cancer8-13, offer many opportunities to apply this technique to BPA and other environmental chemicals.
Those impacts offer opportunities for further investigation of Bisphenol A as an active promoter of disease which works in opposition to currently used pharmaceuticals and chemotherapy compounds.
1. (Nature 532, 166–168 (14 April 2016) doi:10.1038/532166a)
2. Medical devices and articles used in product manufacturing containing bisphenol-A related materials. Available at: www.elsevierbi.com/∼/media/EBD920AE0C5849238635354137B6B082. Accessed May 10, 2016.
3. LaPensee EW, Tuttle TR, Fox SR, Ben-Jonathan N. Bisphenol A at Low Nanomolar Doses Confers Chemoresistance in Estrogen Receptor-α–Positive and –Negative Breast Cancer Cells. Environmental Health Perspectives. 2009;117(2):175-180. doi:10.1289/ehp.11788.
5. RedHill and Fraunhofer collaborate on cancer drug, DDN News, , April 16, 2016, Vol. 12, NO. 4. Available at http://www.ddn-news.com/index.php?newsarticle=10449. Accessed May 10, 2016.
6. RedHill Biopharma Acquires Option for Phase II Pancreatic Cancer Drug RP101. Company news release, Available at http://ir.redhillbio.com/releasedetail.cfm?releaseid=866033. Accessed May 10, 2016.
7. Chen, Zhuo-Jia, et al. “Bisphenol A modulates colorectal cancer protein profile and promotes the metastasis via induction of epithelial to mesenchymal transitions.” Archives of toxicology 89.8 (2015): 1371-1381.
Comments are closed.